Blank Fsis 7234 1 PDF Template
Navigating the requirements for labeling within the food industry presents a myriad of challenges, one of which involves the thorough completion and submission of the FSIS Form 7234-1. This particular form, serving as an application for the approval of labels, markings, or devices, plays a critical role in ensuring compliance with USDA regulations. It encompasses a comprehensive range of information—from the specifics of the product such as type, name, and processing category to the details about labeling space and special claims. Additionally, it requires disclosure of any foreign languages used on the label, along with a translation. For businesses seeking to market their poultry, meat, or egg products effectively while adhering to safety standards, understanding every facet of this form is essential. It meticulously outlines the need for identifying the area of the principal display panel, total labeling space, and the inclusion of any special claims or certifications that might influence consumer choice or dietary restrictions. Moreover, the form acknowledges the importance of protecting certain information from mandatory disclosure under the Freedom of Information Act. Completing this form with accuracy and attention to detail not only facilitates the approval process but also exemplifies a company's commitment to quality and safety in the eyes of both regulators and consumers.
Preview - Fsis 7234 1 Form
According to the Paperwork Reduction Act of 1995, an agency may not conduct or sponsor, and a person is not required to respond to, a collection of information unless it displays a valid OMB control number. The valid OMB control number for this information collection is
U.S. DEPARTMENT OF AGRICULTURE
FOOD SAFETY AND INSPECTION SERVICE
APPLICATION FOR
APPROVAL OF LABELS,
MARKING OR DEVICE
FSIS has determined that information provided in items 11, 15, and 16 is exempt from mandatory disclosure under the Freedom of Information Act 5 U.S.C. 552(b)(4).
APPLICANT: See Page 5 for instructions.
1.AGENT NAME, ADDRESS, TELEPHONE NO. (If using an Agent, complete this block, otherwise leave blank.)
2. FOR USDA USE ONLY
5a. NAME OF PRODUCT
5b. HACCP PROCESS CATEGORY (Select one)
03J: Slaughter - all species
03B: Raw Product - ground
03C: Raw Product - not ground
03D: Thermally processed - commercially sterile
03E: Not heat treated - shelf stable
6a. TYPE OF APPROVAL REQUESTED
SKETCH 
 TEMPORARY
TEMPORARY
EXTENSION OF TEMPORARY
6b. WAS THE LABEL PREVIOUSLY APPROVED? 
 YES ►Date of approval:
YES ►Date of approval:
03F: Heat treated - shelf stable  03G: Fully cooked - not shelf stable
03G: Fully cooked - not shelf stable
03H: Heat treated but not fully cooked - not shelf stable
03I: Product with secondary inhibitors - not shelf stable
NO
Prior approval number: Number of labels on hand: Number of days requested:
|
|
8. Does this label include a |
|
|
Yes |
|
No |
|
|
9. (FOR |
|||||||
|
|
|
|
||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
10. Are there any special claims, guarantees, or foreign language on the label? |
|
|
YES |
|
NO (If yes, check all that apply) |
|
|
|
|||||||
|
|
|
|
|
|
|
|
||||||||||
|
|
|
Allergen Statements |
|
|
|
|
|
|
||||||||
|
|
|
|
|
|
|
|
|
|
|
Other Claims: Specify |
|
|
|
|||
|
|
|
Animal Production/Breed/Raising |
|
|
Grading Terms |
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
Certified/Verified |
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
Guarantees |
|
|
|
|
|
|
|
|
||||
|
|
|
Environmental/Green |
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
|
|
Natural/Organic |
|
|
|
|
|
|
|
|
||||
|
|
|
Export Only Labels w/deviations from Domestic Requirements |
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Foreign Language |
|
|
Nutrition/Health |
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
|
|
|
Geographic/Undefined Style |
|
|
Religious Exemption |
|
|
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
11. NAME AND ADDRESS OF FIRM (Below and between dots) |
|
|
|
|
|
|
12. SIGNATURE OF APPLICANT OR AGENT |
13. DATE |
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
14. (FOR USDA USE ONLY) CONDITIONS APPLYING TO USE OF LABELS OR DEVICE |
|||||
FSIS FORM |
REPLACES FSIS FORM |

FSIS FORM
15. PRODUCT FORMULA
PAGE of
PCT |
|
WEIGHT |
(No Fractions)
|
|
See Continuation Sheet |
TOTAL (Percent must total 100%) |
|
|
|
|

FSIS FORM |
PAGE |
|
of |
|
|
|
|
16. PROCESSING PROCEDURES (Approval of the sketch does not convey approval of the processing procedures)

 See Continuation Sheet
See Continuation Sheet

FSIS FORM |
PAGE |
|
of |
|
|
|
|
||||
|
|
|
|
|
|
CONTINUATION SHEET FOR APPLICATION FOR APPROVAL OF LABELS,
MARKING OR DEVICE (FSIS
PRODUCT NAME:
This sheet is being used for additional information required in Block(s):
INSTRUCTIONS FOR PREPARATION OF FSIS FORM
Note: The following instructions should be typed unless otherwise noted.
A. |
PREPARATION OF APPLICATION |
7b. |
Total available labeling space in square inches for entire package. |
|
Application must be typed or it will be returned without evaluation. |
8. |
|
|
Submit two copies for each label application. |
||
|
|
|
includes a |
B. |
TYPE OF APPROVAL REQUESTED |
9. |
Leave Blank. For |
|
Sketch: Self explanatory. (See 9 CFR 317.4 & 381.132) |
||
|
|
|
|
|
Temporary and Extension of Temporary. Actual label or color litho take off to be used. |
10. |
Special claims, guarantees, or foreign language. Indicate if there are |
|
|
|
are any special claims, guarantees, or foreign language on the label. |
C. |
FOREIGN LANGUAGE |
|
Check all that apply. If Other Claims is selected, indicate specific claim(s) |
|
Labels printed in foreign languages must be accompanied by English language translation. |
|
|
|
|
in space provided. |
|
|
|
|
|
D. |
ASSEMBLY OF APPLICATION |
11. |
Name and Address of Firm. Insert Firm's name and mailing |
|
Application Form, Product Formula, Processing Procedures, Continuation Sheet if applicable, Label, |
|
address. Use 2 letter symbol for State. Show postal zip code. |
|
and any Supporting Documentation Staple with one or as few staples as possible. (Do not use paper |
12 & 13. |
Signature and Date of Applicant or Agent. To be signed and |
|
clips). |
||
|
|
|
dated by the applicant or agent representing the official |
E. |
MAIL COMPLETED APPLICATION TO: |
|
establishment or plant. |
|
|
||
|
USDA, FSIS, OPPD, LPDD |
14. |
|
|
Labeling Distribution Unit |
Leave blank for USDA use only. Conditions Applying to Use of Label or |
|
|
|
||
|
Stop Code 3786, Patriots Plaza III, |
|
Device. (Any condition, modification or remarks applied to the application |
|
1400 Independence Avenue, SW |
|
when approved are conditions governing use of the approved devices.) |
|
Washington, DC |
|
|
The following instructions relate to numbered items on form. |
15. |
Product Formula. List the ingredients by percent or weight in order of |
|
1. |
If using an Agent, provide the company name, address, and telephone number, |
|
their predominance. If product consists of several components, e.g., |
|
a frozen dinner, list each component separately and indicate the |
||
|
otherwise leave blank. |
|
percentage or amount of each component in the product. If additional |
2 & 3. |
Leave blank, for USDA use only. |
|
space is needed, check the box for "Continuation Sheet," and use the |
|
Continuation Sheet. Be sure to include the product name and number |
||
|
|
|
of the block item. Express all ingredients in the same units, i.e., do not |
4. |
Establishment No./Foreign Country (if applicable) - Self Explanatory. |
|
list some in pounds and others in ounces. |
4a. |
Type of Product. Select one product type: Egg, Meat, Poultry, or Other (i.e. Exotic Species, |
|
|
|
Check whether weight or percent is used. It is preferred that |
||
|
|
||
5a. |
Name of Product. Use common or descriptive product name, i.e., "Frankfurter , |
|
percentages be used, and the total must equal 100 percent. If weights |
|
are used, show in pounds, kilograms or grams. (No gallons, pints, |
||
|
Cereal Added" or "Meat Patties in Gravy. (Do not use trade brand names or |
|
|
|
|
cups, teaspoons, etc.) The total must equal the weights of the |
|
|
coined names, such as "Joe's Corn Dogs" or "Joe's Sloppy Joes.") If coined |
|
|
|
|
individual units. (Example: Crust + Cheese + Sauce + Meat = Total |
|
|
names such as "Corn Dogs" are used, also show true product name, such as |
|
|
|
|
new weight of unit.) |
|
|
"Batter Wrapped Wiener." |
|
|
|
|
|
|
5b. |
Provide HACCP process category for the product. See 9 CFR 417.2(b) (1), |
|
DO NOT use fractions. Express as decimals carried to two places, Example: |
|
Example, Heat Treated - shelf stable, Not heat |
|
|
6a & b. |
Type of Approval Requested. If temporary approval or extension, insert |
|
|
|
number of days requested and number of labels on hand. If previous |
|
|
|
approval, attach copy of application and label. Include specific reason(s) |
16. |
Processing Procedures. Poultry Products provide complete |
|
why requesting a temporary or extension and include information required |
||
|
in 9 CFR 317.4(f) (1) or 381.132(f) (1) on the continuation sheet. |
|
processing procedures as required in 9 CFR 381.134. Meat |
|
Be sure to include product name and block item. |
|
Products, provide complete processing procedures as required. |
|
|
|
Note: Approval of the sketch does not convey approval of the |
7a. |
Area of Principal Display Panel (PDP). The PDP is the entire side |
|
processing procedures. If additional space is needed, check the box for |
|
"Continuation Sheet," and use the Continuation Sheet. |
||
of the package to which the label is affixed. See 9 CFR 317.2 (d) |
Be sure to include the product name and number of the block item. |
|
and 381.116 (b). |
||
|
||
|
|
|
FSIS FORM |
|
Form Data
| Fact Name | Detail |
|---|---|
| Form Number and Version | FSIS Form 7234-1 (11/16/2011) |
| Supersedes | Replaces FSIS Form 7234-1 (09/01/2009), which is obsolete. |
| Purpose of Form | Application for Approval of Labels, Marking, or Device |
| OMB Control Number | 0583-0092 |
| Estimated Completion Time | Average 75 minutes per response |
| Authorization | Approved by OMB for web distribution under the Paperwork Reduction Act of 1995 |
| Exemption Clause | Information in items 11, 15, and 16 is exempt from mandatory disclosure under the Freedom of Information Act 5 U.S.C. 552(b)(4) |
| Submission Requirements | Must be typed and submitted in two copies for each label application. |
| Special Instructions | Includes specific instructions on preparing the application, type of approval requested, foreign language labels, and assembly of application |
| Mailing Address | USDA, FSIS, OPPD, LPDD Labeling Distribution Unit, Stop Code 3786, Patriots Plaza III, 8-168, 1400 Independence Avenue, SW, Washington, DC 20250-3700 |
| Regulating Laws for Label Approval | Governed by specific CFR (Code of Federal Regulations) sections such as 9 CFR 317.4 & 381.132 for sketches, and 9 CFR 317.2(d) & 381.116(b) for the Area of Principal Display Panel |
Instructions on Utilizing Fsis 7234 1
After preparing your FSIS 7234-1 form, your application will be reviewed by the USDA Food Safety and Inspection Service. This step is crucial for obtaining approval for your labels, markings, or devices related to meat, poultry, or egg products. Accurately filling out this form ensures a smoother approval process and helps avoid any delays. Below are the detailed steps you should follow to complete the form correctly.
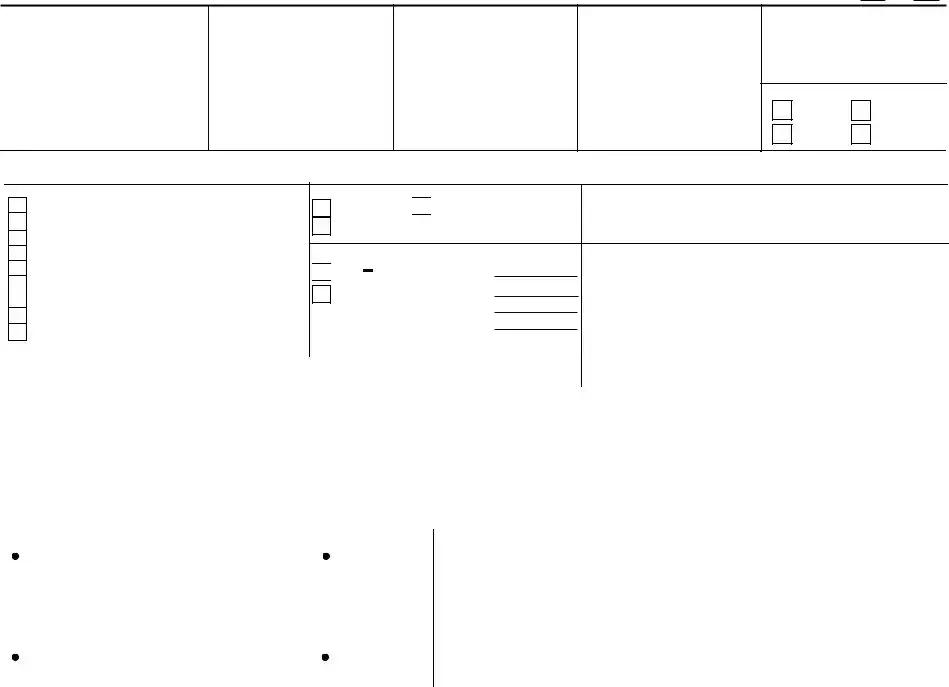
- For items 1 and 2, if you're using an agent, provide the agent's name, address, and telephone number. Leave these items blank if you're not using an agent.
- Items 3 and 9 are for USDA use only. Do not fill these out.
- In item 4, clearly indicate the Establishment Number/Foreign Country, if applicable. This item is straightforward and requires specific information about your establishment.
- For item 4a, select the type of product from the options provided (e.g., Egg, Meat, Poultry, Other).
- In items 5a and 5b, describe the name of your product and select the appropriate HACCP process category that applies.
- Regarding item 6a, specify the type of approval requested (Sketch, Temporary, Extension of Temporary) and if the label was previously approved (6b), provide the approval date and prior approval number.
- Answer "Yes" or "No" to whether your label includes a USDA-AMS Child Nutrition Program CN-Logo in item 8.
- For item 10, indicate whether there are any special claims, guarantees, or foreign languages on the label by checking "Yes" or "No" and specifying the claims, if applicable.
- Item 11 requires the name and address of your firm. Ensure this information is accurate and up-to-date.
- Sign and date the form in items 12 and 13 to authenticate the information provided.
- Leave item 14 blank as it is for USDA use only.
- Item 15: Provide your product's formula including each ingredient's percentage by weight, ensuring that the total percentage equals 100%. Use the Continuation Sheet if additional space is needed.
- For item 16, outline your product's processing procedures in detail. If the space provided is insufficient, use the Continuation Sheet and include the product name and corresponding item block number.
- Measure the area of the Principal Display Panel (7a) and the Total Available Labeling Space for the Entire Package (7b) in square inches, as these dimensions are crucial for label approval.
Once you've completed all relevant parts of the form, assemble your application package, including the application form, product formula, processing procedures, continuation sheet if used, actual label or a color lithograph of the label, and any supporting documentation. Remember to staple the package together securely. Finally, mail the completed application to the address provided in the instructions, ensuring you've included two copies for each label application. Carefully following these steps will facilitate a smoother review process by the USDA.
Obtain Answers on Fsis 7234 1
What is the FSIS 7234-1 form?
The FSIS 7234-1 form, developed by the United States Department of Agriculture (USDA) Food Safety and Inspection Service (FSIS), is an application for the approval of labels, marking, or devices used on meat, poultry, and egg products. It is essential for ensuring that labels are accurate, not misleading, and compliant with federal regulations.
Who needs to fill out the FSIS 7234-1 form?
Companies or individuals who produce meat, poultry, or egg products and intend to sell these products within the United States must complete the form. This includes domestic producers and foreign exporters to the U.S. who need their labeling approved by FSIS before their products can enter the U.S. market.
What information is required on the FSIS 7234-1 form?
The form requires detailed information about the product and the label, including:
- The area of the principal display panel and total available labeling space.
- Type of product (e.g., meat, poultry, egg).
- Name of the product.
- HACCP (Hazard Analysis Critical Control Point) process category.
- Type of approval requested (e.g., sketch, temporary, extension of temporary).
- Whether the label was previously approved.
- Details about any special claims, guarantees, or foreign languages on the label.
- Product formula and processing procedures.
How long does it take to fill out the form?
On average, completing the FSIS 7234-1 form, including time for reviewing instructions, searching existing data sources, gathering and maintaining the needed data, and reviewing the collection of information, is estimated to take about 75 minutes per response.
Is there a valid OMB control number for the form?
Yes, the valid OMB control number for this information collection is 0583-0092. This number indicates that the Office of Management and Budget (OMB) has approved the form and its use.
Are there any parts of the form that are exempt from mandatory disclosure?
Yes, FSIS has determined that information provided in items 11, 15, and 16 is exempt from mandatory disclosure under the Freedom of Information Act (5 U.S.C. 552(b)(4)). This typically includes proprietary information such as detailed product formulas and processing procedures.
Can labels in foreign languages be approved?
Yes, labels printed in foreign languages can be approved. However, they must be accompanied by an English language translation to ensure FSIS can accurately assess the label for compliance with U.S. regulations.
What happens if the application isn't filled out correctly?
If the application is not filled out correctly or completely, it will be returned without evaluation. It is crucial to follow the provided instructions closely and ensure all required information is included before submission.
Where should the completed FSIS 7234-1 form be mailed?
Completed forms should be mailed to: USDA, FSIS, OPPD, LPDD, Labeling Distribution Unit, Stop Code 3786, Patriots Plaza III, 8-168, 1400 Independence Avenue, SW, Washington, DC 20250-3700. This address ensures the application is directed to the appropriate department for evaluation.
Common mistakes
Filling out the FSIS 7234-1 form, an application for the approval of labels, marking, or device, is a crucial process for ensuring that food product labels meet the U.S. Department of Agriculture's standards. However, people often make mistakes in this process. Here are seven common errors to avoid:
Not providing complete information in item 7a (Area of Principal Display Panel) and 7b (Total Available Labeling Space for Entire Package). It's important to accurately calculate and enter the square inches of the principal display panel and the total labeling space of the package. Underestimating or overestimating these areas can lead to non-compliance with USDA standards.
Incorrectly identifying the product type in item 4a. With options including Meat, Poultry, Egg, and Other, accurately identifying the product type is crucial for the application. Selection mistakes can lead to incorrect processing and approval delays.
Forgetting to indicate if the product includes a USDA-AMS Child Nutrition Program Logo in item 8. This detail is pivotal for products that intend to participate in child nutrition programs and must be clearly stated.
Omitting special claims in item 10. If your product labels have special claims, guarantees, or ingredients listed in a foreign language, failing to check the appropriate box under item 10 might lead to issues with label approval.
Leaving the name and address of the firm vague in item 11. The firm's name and complete mailing address, including the correct zip code and State symbol, are mandatory for procedural accuracy and communication purposes.
Failure to include the product formula or processing procedures in items 15 and 16, respectively. These sections are critical for the USDA's assessment of the product's compliance with safety standards. Ignoring these sections or providing incomplete information can significantly delay the approval process.
Not submitting the application in the correct format or without necessary documentation. According to the instructions, the application must be typed and accompanied by all necessary documentation, including the product formula, processing procedures, and any additional requested information. Failure to comply with these instructions can result in the application being returned without evaluation.
Avoiding these common mistakes can streamline the approval process for your food product labels, ensuring they meet the Food Safety and Inspection Service (FSIS) requirements. Make sure to review your application thoroughly before submission.
Documents used along the form
When handling the FSIS Form 7234-1, it's vital to recognize its role as a cornerstone in the broader tapestry of regulatory documentation related to food safety and labeling in the United States. This form, tailored for the submission of labels, markings, or devices for approval by the Food Safety and Inspection Service, integrates into a network of required documentation, ensuring that food products meet stringent safety and labeling standards before they reach consumers. Understanding additional documents often used alongside FSIS Form 7234-1 will elucidate the comprehensive process behind food safety and labeling approval.
- Ingredient Statement: This document lists all the ingredients contained in the product, presented in descending order by weight. It is crucial for verifying the compliance of the product's formulation with safety and labeling regulations.
- Nutrition Facts Label: A standardized label that provides detailed information about the nutritional content of the product, including calories, macronutrients, vitamins, and minerals. It is essential for ensuring that the product meets the nutritional labeling requirements set by the FDA.
- Manufacturer's Warranty: This document guarantees the product's compliance with specified standards and regulations. It may be required to support claims made on the labeling, such as "organic" or "non-GMO."
- Allergen Declaration: A critical document that lists all potential allergens present in the product, ensuring compliance with labeling laws that mandate the disclosure of common allergens to protect consumers with allergies.
- USDA Grant of Inspection: This certificate denotes that the facility producing the product complies with USDA hygiene and safety standards, necessary for the approval of meat, poultry, and egg products.
- HACCP Plan: A document detailing the Hazard Analysis and Critical Control Point (HACCP) plan of the establishment, illustrating the steps taken to ensure food safety throughout the production process.
- Label Registration Proof: Proof that the product's label has been registered with the appropriate regulatory body, often required for specific claims or certification marks.
- Country of Origin Certificate: This certificate, required for imported products, verifies the country of origin, ensuring compliance with import regulations and labeling requirements.
Together with the FSIS Form 7234-1 application for label, marking, or device approval, these documents form an interlocking framework that rigorously secures the safety and integrity of food products. This meticulous process, although complex, underscores the United States' commitment to maintaining one of the safest food supplies in the world. Understanding the breadth and interrelation of these documents not only demystifies the regulatory landscape but also highlights the extensive efforts undertaken to protect consumers, ensuring that they have access to safe and accurately labeled food products.
Similar forms
The FDA Food Facility Registration Form is similar because it requires detailed information about the facility and its products, similar to the FSIS 7234-1 form that collects specific details about the meat, poultry, or egg product being labeled.
The Nutrition Facts Label submission forms used by companies to ensure their food products' labels comply with federal regulations, like the FSIS 7234-1 form, ensure compliance with USDA labeling requirements.
The TTB COLAs (Certificate of Label Approval) for alcohol beverages, which, like the FSIS 7234-1, need federal approval before labeling certain products to ensure that they meet defined standards.
The EPA Pesticide Labeling Approval Form, which requires detailed product and usage information for approval, reflects the FSIS 7234-1 form's need for specific product details for regulatory compliance.
The USDA Organic Certification Application, which requires detailed descriptions of the product and production processes, shares similarities in content and the level of detail required with the FSIS 7234-1 form.
US Customs & Border Protection (CBP) Product Labeling Requirement Forms for importing goods, similarly demand extensive product information to ensure compliance with U.S. standards, akin to the detailed labeling requirements stipulated in the FSIS 7234-1 form.
The FCC Equipment Authorization Application for RF Devices, like the FSIS 7234-1, requires specific product information to assess compliance with regulations, ensuring public safety.
The USDA APHIS Petition for Nonregulated Status regarding genetically engineered plants, which also requires exhaustive product information for regulatory assessment, sharing the FSIS 7234-1's intent to scrutinize products closely for public safety.
NOAA Seafood Inspection Program Application Forms, which ascertain compliance with seafood safety standards, align with the FSIS 7234-1's goal to ensure food products are safely labeled and marketed.
The USDA AMS Commodity Procurement Program Applications that demand detailed product descriptions and specifications, resembling the FSIS 7234-1 form's requirement for thorough product information for label approval.
Dos and Don'ts
When preparing to fill out the FSIS Form 7234-1, it's important to approach the task with careful attention to detail and a thorough understanding of the requirements. The following are recommendations on what to do and what to avoid for a successful submission.
What You Should Do:
- Read all instructions thoroughly before starting to fill out the form. Understand the specific requirements for each section to ensure accuracy.
- Provide accurate and detailed information in every section, particularly when listing product formulas and processing procedures. This clarity helps in avoiding delays in the approval process.
- Use English language translations for labels in foreign languages, ensuring that all information is accessible and clear to the reviewing agency.
- Include two copies of each label application as specified, to comply with submission guidelines and facilitate the review process.
- Ensure your application is typed, as handwritten applications may be returned without evaluation.
What You Shouldn't Do:
- Avoid submitting incomplete forms. Review each section carefully to ensure that no required information is missing.
- Do not overlook the need for a signature. A missing signature from the applicant or agent can result in the rejection of the application.
- Refrain from using trade brand names or coined names without also providing a common or descriptive product name, as this can lead to confusion.
- Avoid mixing units of measurement in the product formula section. Consistency in using percentages or weight units is crucial for clarity.
- Do not staple documents excessively. Use one staple or as few as possible, as instructed, to keep the application neat and manageable.
By following these guidelines, applicants can streamline the process of obtaining approval for labels, marking, or devices from the FSIS. Attention to detail and adherence to the procedural requirements are key to a successful application.
Misconceptions
Only meat products need to be labeled: This is inaccurate. The FSIS Form 7234-1 is applicable for the labeling of various products including poultry, egg products, and other related items, not just meat.
Submission is optional: It's a common misconception that submitting this form is optional. In truth, for approval of labels, markings, or devices to be used in products regulated by the USDA, submission of this form is mandatory for compliance.
Approval is immediate: Some believe that approval of labels is immediate upon submission. However, the review process involves several steps, including compliance checks and possible revisions, which means approval is not instantaneous.
The form is only for domestic products: The form is equally important for both domestic and imported products requiring USDA inspection and approval. It ensures that foreign products meet the same standards as those produced domestically.
Language requirements are flexible: All labels must include English language translations, especially if they contain foreign language elements. This requirement ensures that all information is accessible to regulatory authorities and consumers.
Sketches are the final step: Submitting a sketch of the label is just a part of the application process. Approval of the sketch does not equate to final approval as processing procedures and other details must also be approved.
All sections must be completed: Sections labeled "FOR USDA USE ONLY" should be left blank by the applicant. Completing these sections can cause confusion and delay the approval process.
Labels previously approved elsewhere are exempt: Even if a label has been approved by another regulatory agency, it must still undergo the FSIS review process. The standards and requirements may differ across agencies.
Any label size is permissible: The form requires specifics about the area of the principal display panel and the total available labeling space. These details matter, as they ensure that the label meets visibility and readability standards set by the USDA.
Agents are unnecessary: While not mandatory, an agent can act on behalf of the applicant, facilitating the process especially for firms that may not have the expertise or time to handle the application process directly. Providing agent details when applicable can be beneficial.
Key takeaways
When filling out and using the FSIS Form 7234-1, it is essential to pay close attention to detail and follow specific guidelines laid out by the U.S. Department of Agriculture (USDA) Food Safety and Inspection Service (FSIS). Below are critical takeaways that applicants should bear in mind:
- The form requires detailed information about the product and the applicant, including the type of product, establishment number, and if applicable, foreign country information. It's crucial that all provided information is accurate and complete to avoid processing delays.
- The form includes sections for specifying the area of the principal display panel in square inches and the total available labeling space for the entire package. This information helps in determining the suitability of the label size for the product package.
- Applicants can request different types of approval, such as sketch, temporary, and extension of temporary approvals, highlighting the form's flexibility to accommodate various labeling needs.
- If the label encompasses any special claims, guarantees, or foreign language text, it is mandatory to indicate this on the form. This requirement ensures that all label claims are properly reviewed and approved in compliance with USDA guidelines.
- For products that include a USDA-AMS Child Nutrition Program CN-Logo, it’s necessary to indicate this on the form. This inclusion aids in the recognition and verification of products participating in national nutrition programs.
- The form must be accompanied by an English translation if it includes labels printed in foreign languages, ensuring clear understanding and evaluation of the label content by the FSIS.
- Before mailing the completed application, it is essential to assemble the application form, product formula, processing procedures, any continuation sheet if required, the label, and any supporting documentation, and staple them together as instructed. This proper assembly aids in the efficient review and processing of the application.
Sending the complete and correctly assembled application package to the correct address expedites the review process, ensuring that your labeling or marking endeavors comply with federal regulations.
Popular PDF Forms
Do C Corps Issue K-1 - The Schedule K-1 1120-S is more than just a tax document; it's a reflection of the symbiotic relationship between an S corporation and its shareholders, underlined by shared financial successes and obligations.
Lost License Plate Ct - The form cautions that temporary plates may not be acknowledged by other states.
Transmittal Form Meaning - Optimizes project coordination by detailing the delivery of documents such as shop drawings and samples.
